Efficacy data in 2L DTC CABOMETYX® (cabozantinib) monotherapy
In patients who are RAI-R or ineligible and progressed on a prior VEGFR-targeted therapy1
On This Page:
Meaningful results led to early unblinding in the COSMIC-311 trial2
COSMIC-311 was a phase 3, multicenter, randomized, double-blind, placebo-controlled trial.1,3
|
2:1 randomization |
||||
|---|---|---|---|---|
|
||||
|
Treatment until disease progression |
||||
|
UNBLINDED TRIAL‡ |
Cabozantinib 60 mg |
|||
|
Post-treatment follow-up§ |
||||
The multiple primary efficacy outcome measures assessed ORR in the first 100 patients (OITT) after 6 months of enrollment and PFS in all patients randomly assigned (ITT). The study was designed such that it would be considered positive if either of the primary endpoints were met. Median follow-up was 6.2 months (IQR: 3.4–9.2) for the ITT population and 8.9 months (IQR: 7.1–10.5) for the OITT population. Median duration of treatment exposure in the safety population was 4.4 months (IQR: 2.1-7.3) for the CABOMETYX patients and 2.3 months (IQR: 1.6-5.6) for the placebo group. An updated analysis, with median follow-up of 10.1 months, evaluated a total of 258 randomized patients.1,3,4
- *
-
BSC included dose modification and dose interruption. After unblinding, BSC included potentially receiving open-label cabozantinib.1,3
- †
-
Tumor response and progression were assessed by MRI or CT. Tumor assessments were performed every 8 weeks after randomization for 12 months, then every 12 weeks thereafter.3
- ‡
-
An IDMC for COSMIC-311 recommended ending enrollment and unblinding sites and patients based on cabozantinib reducing the risk of disease progression or death by 78% (HR=0.22; 95% CI: 0.14-0.35; P<0.0001).2
Multiple primary efficacy outcome measures1
-
PFS
- ORR
Stratification factors1
-
Age ≤65 years
-
Prior lenvatinib
Baseline characteristics in ITT population1,3
| Total Population | ||
| Median age, years (range) ECOG 0 ECOG 1 |
65 (31-85) 46% 54% |
|
| CABOMETYX | Placebo | |
| One previous VEGFR TKI Two previous VEGFR TKIs |
73% (91/125) 27% (34/125) |
77% (48/62) 23% (14/62) |
73% of patients received CABOMETYX after one previous systemic therapy3§
- §
-
Updated analysis (N=258).1

CABOMETYX delivered a significant benefit in the primary PFS analysis1
Median PFS was not reached in the primary analysis (n=125, 95% CI: 5.7-NE) vs PFS of 1.9 months with placebo (n=62, 95% CI: 1.8-3.6); HR=0.22 (95% CI: 0.14-0.35); P<0.0001.
Early and sustained separation demonstrated at updated analysis–with a median PFS of 11 months1
Updated analysis
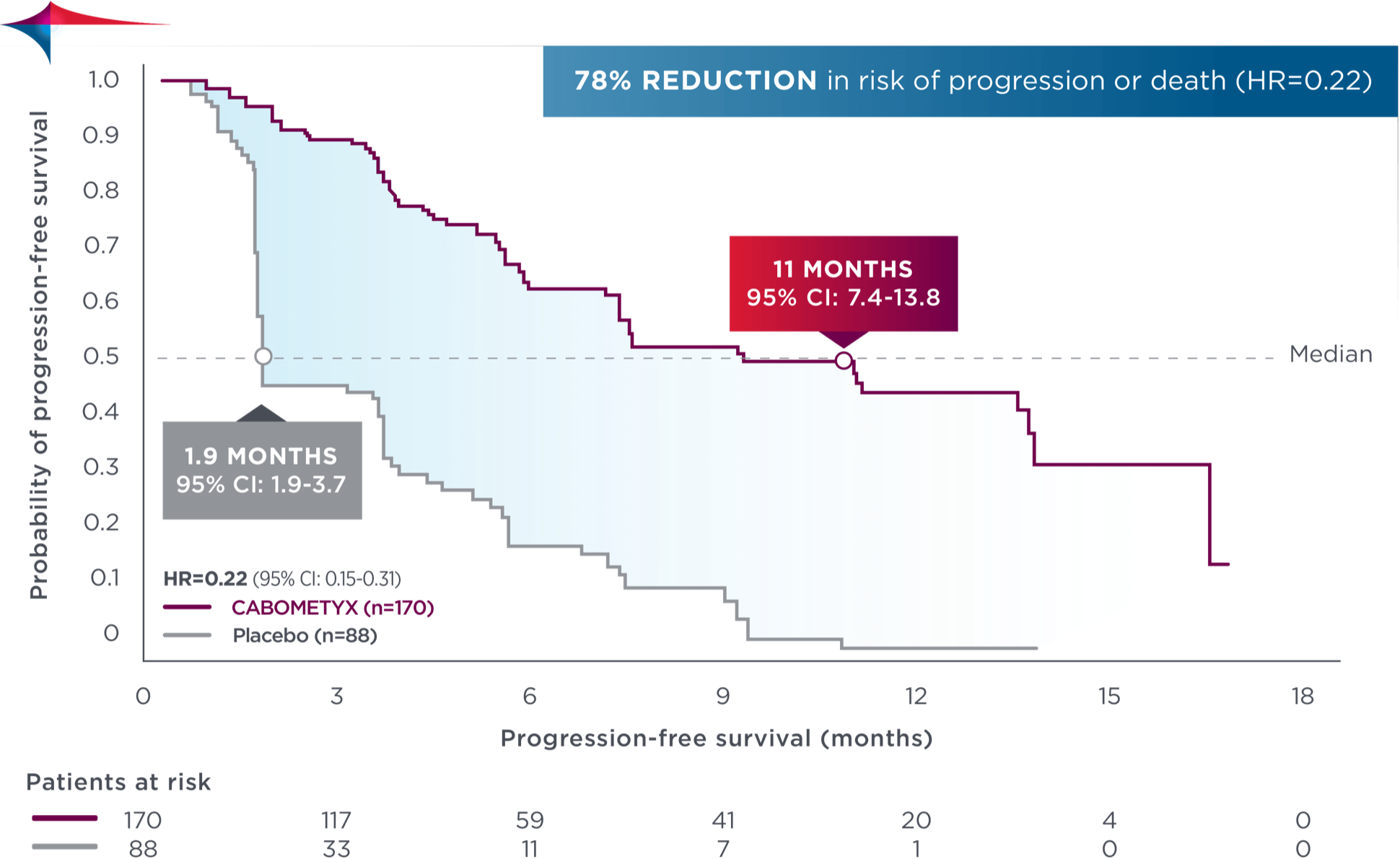
No formal statistical testing was conducted at the time of the updated analysis.
78% reduction in risk of progression or death in both primary and updated analyses
Tumor response observed at the OITT analysis3
In the COSMIC-311 trial, the ORR did not reach a prespecified endpoint for statistical significance (critical P value=0.01). Data below cover tumor response information collected, inclusive of ORR.1,3
|
CABOMETYX |
Placebo |
|
|
ORR, % (95% CI)‖¶ |
15 (7-26) |
0 (0-11) |
- ‖
-
P=0.0281.1
- ¶
-
All responses confirmed were partial responses.3
- **
-
Stable disease is defined as neither sufficient shrinkage to qualify for partial response nor sufficient increase to qualify for PD.5
Stable disease may reflect the natural history of disease rather than any effect of the drug. - ††
-
Disease control rate was defined as the percentage of patients with a complete response, partial response, or stable disease, as measured by RECIST v1.1.3
Descriptive analysis
Reduction in target lesions from baseline3
Of the 100 OITT patients, 89 were evaluable (n=58 in the CABOMETYX arm, n=31 in the placebo arm).
76% of evaluable patients experienced tumor shrinkage with CABOMETYX vs 29% with placebo‡‡
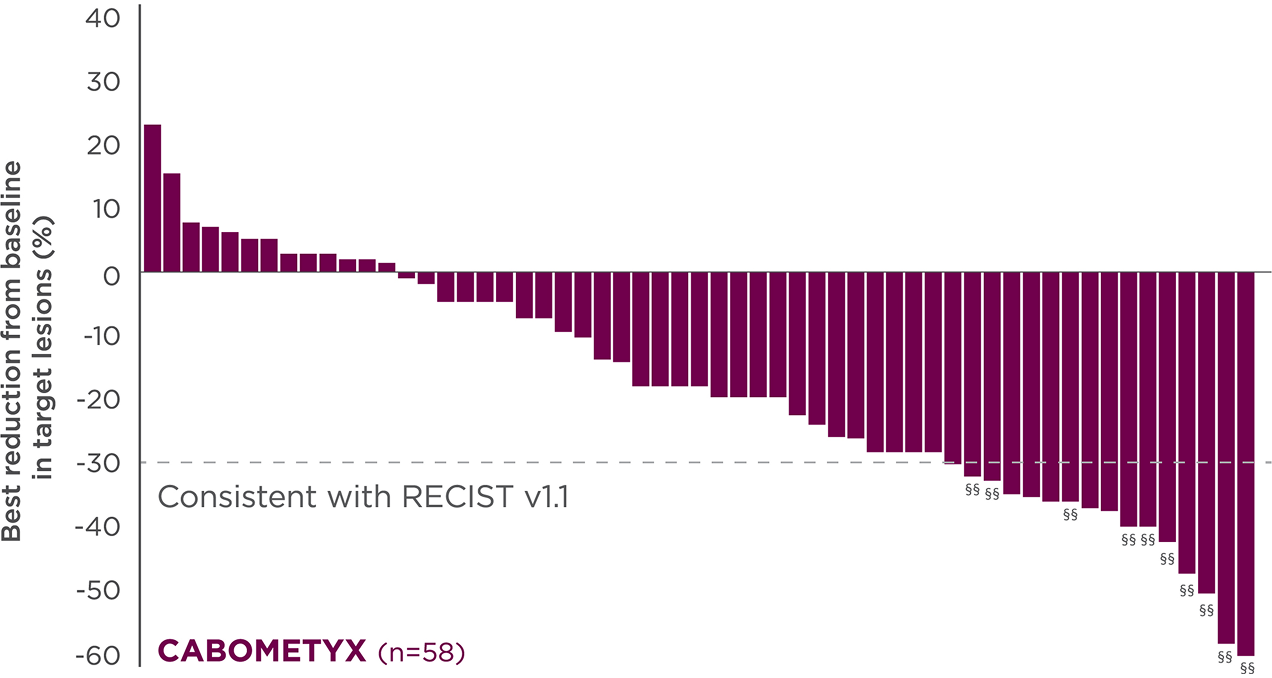

No formal statistical testing was conducted for the tumor assessment waterfall plots; interpret data with caution.
Best reduction is maximum reduction in sum of diameters of target lesions. Patients with target lesion at baseline and at least 1 on-treatment tumor assessment are included. Some patients are not represented due to lack of evaluable postbaseline assessment, censoring before first evaluable postbaseline assessment, lack of target lesions, and/or incomplete or inevaluable target-lesion assessment. Data from time points after the first date of any of the censoring events defined for the primary PFS analysis were excluded.6
- ‡‡
-
Each vertical line represents 1 patient.3
- §§
-
Indicates confirmed partial responses per investigators.3

National Comprehensive Cancer Network® (NCCN®)
Cabozantinib (CABOMETYX) has a Category 1 recommendation for patients with RAI-R papillary thyroid cancerIIII that has progressed following VEGFR-targeted therapy¶¶
-
NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
NCCN Category 1: Based upon high-level evidence (≥1 randomized phase 3 trials or high-quality, robust meta-analyses), there is uniform NCCN consensus (≥85% support of the Panel) that the intervention is appropriate. - ||||
-
Locally recurrent, advanced, and/or metastatic.
- ¶¶
-
If progression after lenvatinib and/or sorafenib.
2L=second-line; BIRC=blinded independent radiology committee; BSC=best supportive care; CI=confidence interval; CT=computed tomography; DTC=differentiated thyroid cancer; ECOG=Eastern Cooperative Oncology Group performance status; HR=hazard ratio; IDMC=independent data-monitoring committee; IQR=interquartile range; ITT=intent to treat; MRI=magnetic resonance imaging; NE=not estimable; OITT=ORR intent to treat; ORR=overall response rate; PD=progressive disease; PFS=progression-free survival; QD=every day; RAI-R=radioactive iodine-refractory; RECIST=Response Evaluation Criteria in Solid Tumors; SoD=sum of diameters; TKI=tyrosine kinase inhibitor; VEGFR=vascular endothelial growth factor receptor.
References:
- CABOMETYX® (cabozantinib) Prescribing Information. Exelixis, Inc.
- Exelixis press release. December 21, 2020. https://ir.exelixis.com/news-releases/news-release-details/exelixis-announces-cabozantinib-significantly-improved
- Brose MS, Robinson B, Sherman SI, et al. Cabozantinib for radioiodine-refractory differentiated thyroid cancer (COSMIC-311): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2021;22(8):1126-1138. doi:10.1016/S1470-2045(21)00332-6
- European Medicines Agency: Committee for Medicinal Products for Human Use (CHMP). Assessment report: CABOMETYX. September 2018. Accessed September 15, 2022.
- Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228-247.
- Data on file. Exelixis, Inc.
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Thyroid Cancer V.1.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed March 27, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org.

