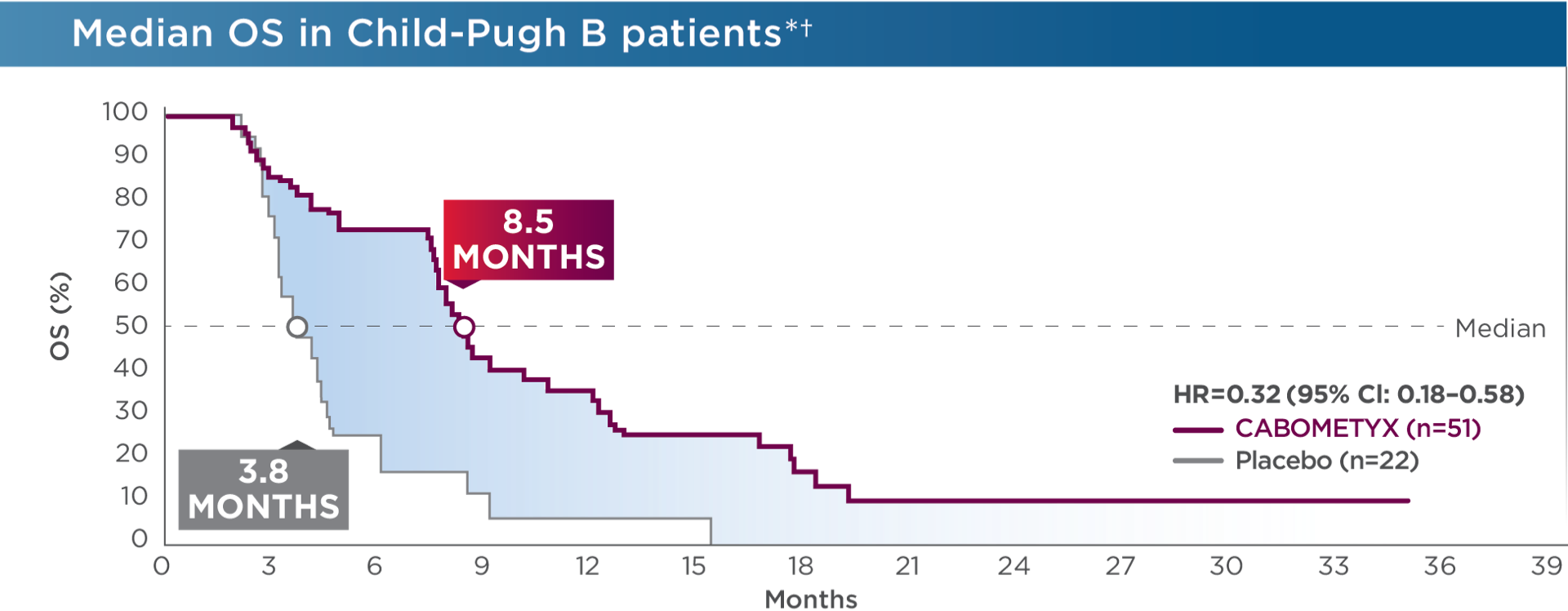
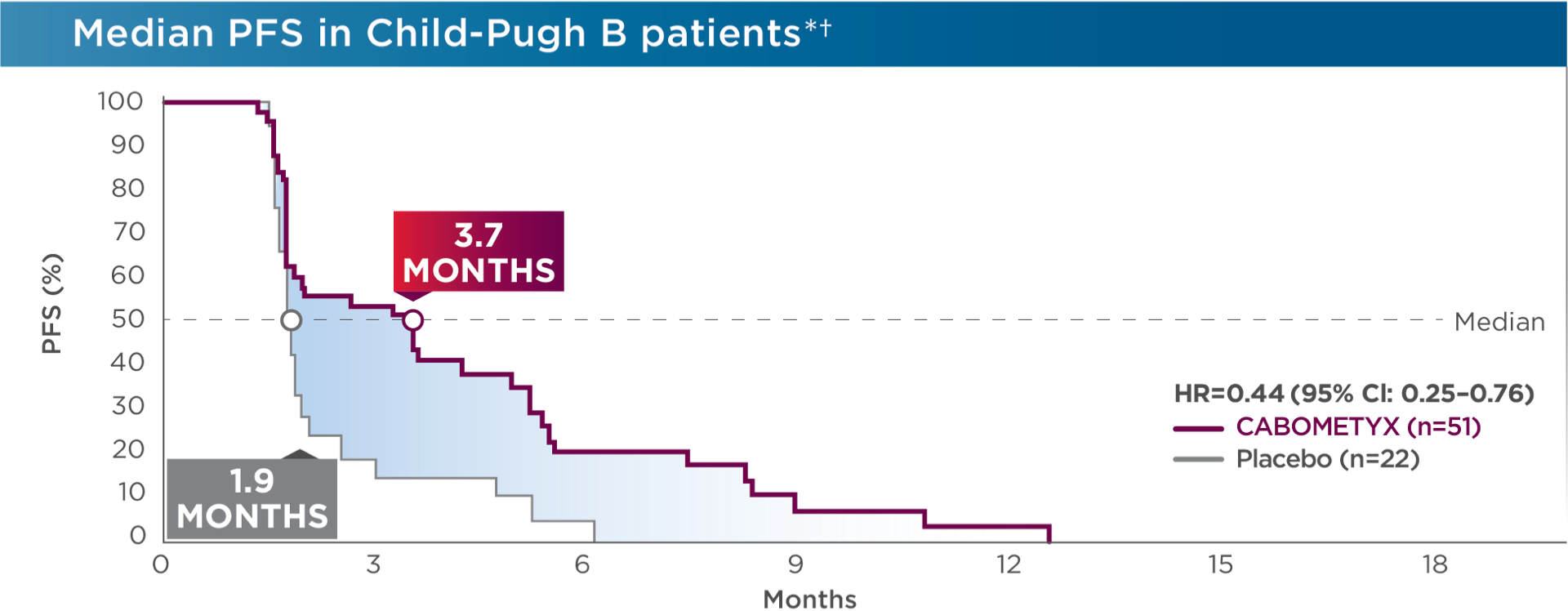
Child-Pugh B: Retrospective exploratory subgroup analysis
OS and PFS were assessed in CELESTIAL patients who progressed from Child-Pugh A to Child-Pugh B status by week 81,2
- *
-
The observed outcomes should be interpreted with caution because of the relatively small size of the Child-Pugh B subgroup.2 No statistical procedure was employed for controlling type I error. Results should be hypothesis generating.3
- †
-
At the time of the first Child-Pugh assessment at Week 8 after randomization by the investigator, 51/470 patients in the CABOMETYX arm and 22/237 patients in the placebo arm had Child-Pugh B cirrhosis (ie, Child-Pugh B subgroup). Available values for the central laboratory-determined Child-Pugh B score points included Child-Pugh B7 (n=26 CABOMETYX arm, 11 placebo), B8 (11 CABOMETYX arm, 3 placebo), and B9 (3 CABOMETYX arm, 5 placebo).2 Child-Pugh grading was investigator assessed and scoring was by Exelixis, Inc. (study sponsor); some discrepancies between grading and scoring results existed.
Select safety results in the Child-Pugh B subgroup2
|
Patients (%) |
||||||
|
CABOMETYX |
Placebo |
|||||
|
All Grades |
Grade 3-4 |
All Grades |
Grade 3-4 |
|||
|
All-causality AR |
||||||
|
Any reaction |
51 (100) |
36 (71) |
22 (100) |
13 (59) |
||
|
Fatigue |
29 (57) |
10 (20) |
9 (41) |
4 (18) |
||
|
Ascites |
17 (33) |
7 (14) |
12 (55) |
5 (23) |
||
|
AST increased |
11 (22) |
7 (14) |
2 (9.1) |
1 (4.5) |
||
|
Thrombocytopenia |
11 (22) |
6 (12) |
0 (0) |
0 (0) |
||
|
Anemia |
6 (12) |
5 (9.8) |
5 (23) |
4 (18) |
||
|
Blood bilirubin increased |
11 (22) |
5 (9.8) |
3 (14) |
0 (0) |
||
|
Dyspnea |
10 (20) |
5 (9.8) |
7 (32) |
0 (0) |
||
|
Blood ALP increased |
4 (7.8) |
4 (7.8) |
0 (0) |
0 (0) |
||
|
Hypertension |
9 (18) |
4 (7.8) |
0 (0) |
0 (0) |
||
|
PPE |
15 (29) |
4 (7.8) |
1 (4.5) |
0 (0) |
||
|
Platelet count decreased |
6 (12) |
4 (7.8) |
0 (0) |
0 (0) |
||
|
Portal vein thrombosis |
4 (7.8) |
4 (7.8) |
0 (0) |
0 (0) |
||
|
Pulmonary embolism |
4 (7.8) |
4 (7.8) |
0 (0) |
0 (0) |
||
|
Asthenia |
12 (24) |
3 (5.9) |
3 (14) |
0 (0) |
||
|
Decreased appetite |
30 (59) |
3 (5.9) |
5 (23) |
0 (0) |
||
|
Diarrhea |
24 (47) |
3 (5.9) |
6 (27) |
1 (4.5) |
||
|
General physical health deterioration |
5 (9.8) |
3 (5.9) |
2 (9.1) |
2 (9.1) |
||
|
Hepatic encephalopathy |
4 (7.8) |
3 (5.9) |
0 (0) |
0 (0) |
||
|
Hyperbilirubinemia |
4 (7.8) |
3 (5.9) |
1 (4.5) |
0 (0) |
||
|
Nausea |
23 (45) |
3 (5.9) |
6 (27) |
0 (0) |
||
|
Pain |
3 (5.9) |
3 (5.9) |
0 (0) |
0 (0) |
||
|
Pneumonia |
4 (7.8) |
3 (5.9) |
1 (4.5) |
0 (0) |
||
|
Abdominal pain |
11 (22) |
2 (3.9) |
10 (45) |
3 (14) |
||
|
Hepatic failure |
3 (5.9) |
1 (2.0) |
3 (14) |
3 (14) |
||
|
Sepsis |
1 (2.0) |
1 (2.0) |
2 (9.1) |
2 (9.1) |
||
|
Additional reactions of interest |
||||||
|
ALT increased |
7 (14) |
2 (3.9) |
1 (4.5) |
0 (0) |
||
|
Hyponatremia |
5 (9.8) |
2 (3.9) |
0 (0) |
0 (0) |
||
|
Neutrophil count decreased |
2 (3.9) |
1 (2.0) |
0 (0) |
0 (0) |
||
|
Hypoalbuminemia |
17 (33) |
1 (2.0) |
2 (9.1) |
0 (0) |
||
|
Chronic hepatic failure |
0 (0) |
0 (0) |
1 (4.5) |
0 (0) |
||
|
61% |
Dose reductions with CABOMETYX2,3 |
18% |
Discontinuations with CABOMETYX due to treatment-related ARs2 |
Patients who progressed to Child-Pugh B received a median average daily dose of 37 mg2
ALP=alkaline phosphatase; ALT=alanine aminotransferase; AST=aspartate aminotransferase; CI=confidence interval; HR=hazard ratio; OS=overall survival; PFS=progression-free survival; PPE=palmar-plantar erythrodysesthesia.
References:
- CABOMETYX® (cabozantinib) Prescribing Information. Exelixis, Inc.
- El-Khoueiry AB, Meyer T, Cheng AL, et al. Safety and efficacy of cabozantinib for patients with advanced hepatocellular carcinoma who advanced to Child–Pugh B liver function at study week 8: a retrospective analysis of the CELESTIAL randomized controlled trial. BMC Cancer. 2022;22(1):377.
- Data on file. Exelixis, Inc.

