IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
Hemorrhage: CABOMETYX can cause severe and fatal hemorrhages. The incidence of Grade 3-5 hemorrhagic events was 5% in CABOMETYX patients in RCC, HCC, and DTC studies. Discontinue CABOMETYX for Grade 3-4 hemorrhage and before surgery. Do not administer to patients who have a recent history of hemorrhage, including hemoptysis, hematemesis, or melena.
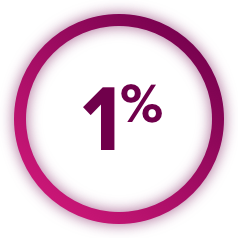
Perforations and Fistulas: Fistulas, including fatal cases, and gastrointestinal (GI) perforations, including fatal cases, each occurred in 1% of CABOMETYX patients. Monitor for signs and symptoms, and discontinue CABOMETYX in patients with Grade 4 fistulas or GI perforation.
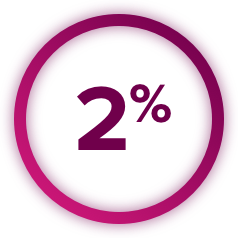
Thromboembolic Events: CABOMETYX can cause arterial or venous thromboembolic events. Venous thromboembolism occurred in 7% (including 4% pulmonary embolism) and arterial thromboembolism in 2% of CABOMETYX patients. Fatal thrombotic events have occurred. Discontinue CABOMETYX in patients who develop an acute myocardial infarction or serious arterial or venous thromboembolic events.
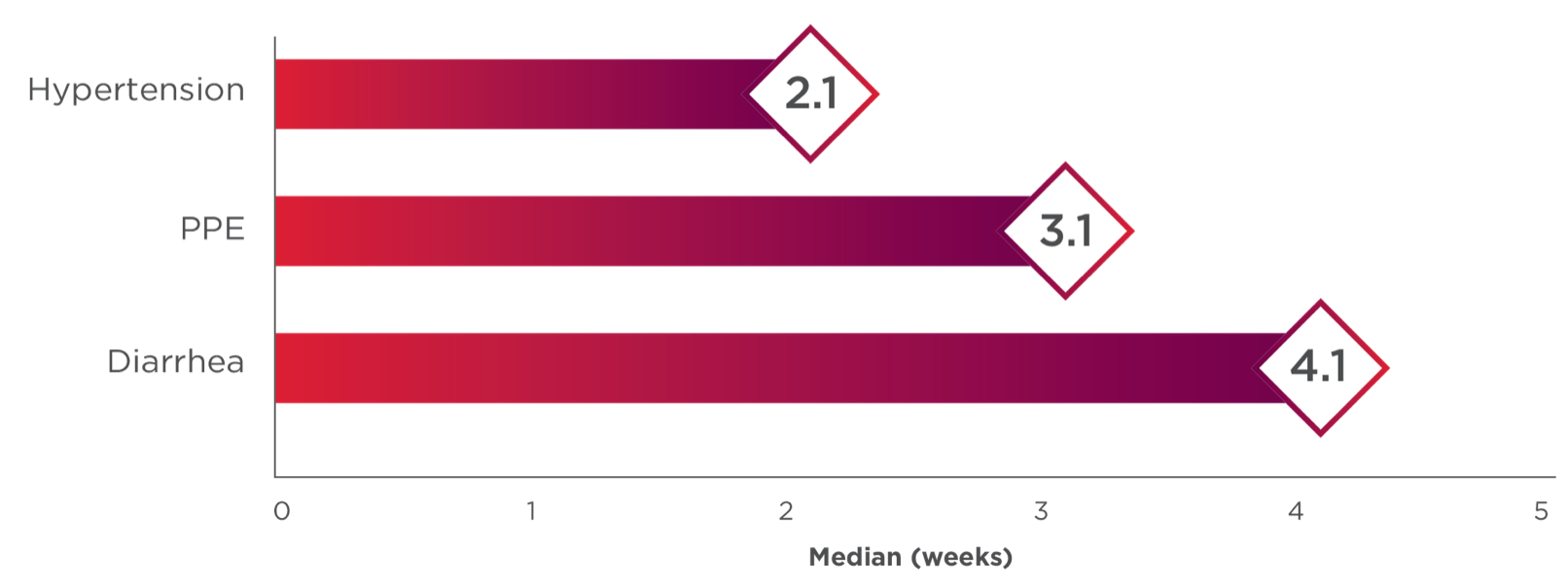
Hypertension and Hypertensive Crisis: CABOMETYX can cause hypertension, including hypertensive crisis. Hypertension was reported in 37% (16% Grade 3 and <1% Grade 4) of CABOMETYX patients. In CABINET (n=195), hypertension occurred in 65% (26% Grade 3) of CABOMETYX patients. Do not initiate CABOMETYX in patients with uncontrolled hypertension. Monitor blood pressure regularly during CABOMETYX treatment. Withhold CABOMETYX for hypertension that is not adequately controlled; when controlled, resume at a reduced dose. Permanently discontinue CABOMETYX for severe hypertension that cannot be controlled with antihypertensive therapy or for hypertensive crisis.
Cardiac Failure: CABOMETYX can cause severe and fatal cardiac failure. Cardiac failure occurred in 0.5% of patients treated with CABOMETYX as a single agent, including fatal cardiac failure in 0.1% of patients. Consider baseline and periodic evaluations of left ventricular ejection fraction. Monitor for signs and symptoms of cardiovascular events. Withhold and resume at a reduced dose upon recovery or permanently discontinue depending on the severity.
Diarrhea: CABOMETYX can cause diarrhea and it occurred in 62% (10% Grade 3) of treated patients. Monitor and manage patients using antidiarrheals as indicated. Withhold CABOMETYX until improvement to ≤ Grade 1; resume at a reduced dose.
Palmar-Plantar Erythrodysesthesia (PPE): CABOMETYX can cause PPE and it occurred in 45% of treated patients (13% Grade 3). Withhold CABOMETYX until PPE resolves or decreases to Grade 1 and resume at a reduced dose for intolerable Grade 2 PPE or Grade 3 PPE.
Hepatotoxicity: CABOMETYX in combination with nivolumab in RCC can cause hepatic toxicity with higher frequencies of Grades 3 and 4 ALT and AST elevations compared to CABOMETYX alone. With the combination of CABOMETYX and nivolumab, Grades 3 and 4 increased ALT or AST were seen in 11% of patients. Monitor liver enzymes before initiation of treatment and periodically. Consider more frequent monitoring as compared to when the drugs are administered as single agents. Consider withholding CABOMETYX and/or nivolumab, initiating corticosteroid therapy, and/or permanently discontinuing the combination for severe or life-threatening hepatotoxicity.
Adrenal Insufficiency: CABOMETYX in combination with nivolumab can cause primary or secondary adrenal insufficiency. Adrenal insufficiency occurred in 4.7% (15/320) of patients with RCC who received CABOMETYX with nivolumab, including Grade 3 (2.2%), and Grade 2 (1.9%) adverse reactions. Withhold CABOMETYX and/or nivolumab and resume CABOMETYX at a reduced dose depending on severity.
Proteinuria: Proteinuria was observed in 8% of CABOMETYX patients. Monitor urine protein regularly during CABOMETYX treatment. For Grade 2 or 3 proteinuria, withhold CABOMETYX until improvement to ≤ Grade 1 proteinuria; resume CABOMETYX at a reduced dose. Discontinue CABOMETYX in patients who develop nephrotic syndrome.
Osteonecrosis of the Jaw (ONJ): CABOMETYX can cause ONJ and it occurred in <1% of treated patients. Perform an oral examination prior to CABOMETYX initiation and periodically during treatment. Advise patients regarding good oral hygiene practices. Withhold CABOMETYX for at least 3 weeks prior to scheduled dental surgery or invasive dental procedures. Withhold CABOMETYX for development of ONJ until complete resolution; resume at a reduced dose.
Impaired Wound Healing: CABOMETYX can cause impaired wound healing. Withhold CABOMETYX for at least 3 weeks prior to elective surgery. Do not administer for at least 2 weeks after major surgery and until adequate wound healing. The safety of resumption of CABOMETYX after resolution of wound healing complications has not been established.
Reversible Posterior Leukoencephalopathy Syndrome (RPLS): CABOMETYX can cause RPLS. Perform evaluation for RPLS and diagnose by characteristic finding on MRI any patient presenting with seizures, headache, visual disturbances, confusion, or altered mental function. Discontinue CABOMETYX in patients who develop RPLS.
Thyroid Dysfunction: CABOMETYX can cause thyroid dysfunction, primarily hypothyroidism, and it occurred in 19% of treated patients (0.4% Grade 3). Assess for signs of thyroid dysfunction prior to the initiation of CABOMETYX and monitor for signs and symptoms during treatment.
Hypocalcemia: CABOMETYX can cause hypocalcemia, with the highest incidence in DTC patients. Based on the safety population, hypocalcemia occurred in 13% of CABOMETYX patients (2% Grade 3 and 1% Grade 4).
Monitor blood calcium levels and replace calcium as necessary during treatment. Withhold and resume CABOMETYX at a reduced dose upon recovery or permanently discontinue CABOMETYX depending on severity.
Embryo-Fetal Toxicity: CABOMETYX can cause fetal harm. Advise pregnant women of the potential risk to a fetus and advise females of reproductive potential to use effective contraception during treatment with CABOMETYX and for 4 months after the last dose.
ADVERSE REACTIONS
The most common (≥20%) adverse reactions are:
CABOMETYX as a single agent: diarrhea, fatigue, PPE, decreased appetite, hypertension, nausea, vomiting, weight decreased, and constipation.
CABOMETYX in combination with nivolumab: diarrhea, fatigue, hepatotoxicity, PPE, stomatitis, rash, hypertension, hypothyroidism, musculoskeletal pain, decreased appetite, nausea, dysgeusia, abdominal pain, cough, and upper respiratory tract infection.
DRUG INTERACTIONS
Strong CYP3A4 Inhibitors: If coadministration with strong CYP3A4 inhibitors cannot be avoided, reduce the CABOMETYX dosage. Avoid grapefruit or grapefruit juice.
Strong or Moderate CYP3A4 Inducers: If coadministration with strong or moderate CYP3A4 inducers cannot be avoided, increase the CABOMETYX dosage. Avoid St. John’s wort.
USE IN SPECIFIC POPULATIONS
Lactation: Advise women not to breastfeed during CABOMETYX treatment and for 4 months after the final dose.
Hepatic Impairment: In patients with moderate hepatic impairment, reduce the CABOMETYX dosage. Avoid CABOMETYX in patients with severe hepatic impairment.
Pediatric Use: Physeal widening has been observed in children with open growth plates when treated with CABOMETYX. Physeal and longitudinal growth monitoring is recommended in children (12 years and older) with open growth plates. Consider interrupting or discontinuing CABOMETYX if abnormalities occur. The safety and effectiveness of CABOMETYX in pediatric patients less than 12 years of age have not been established.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.FDA.gov/medwatch or call 1-800-FDA-1088.