Previously treated epNET efficacy data
On This Page:
CABOMETYX doubled median PFS in epNET1
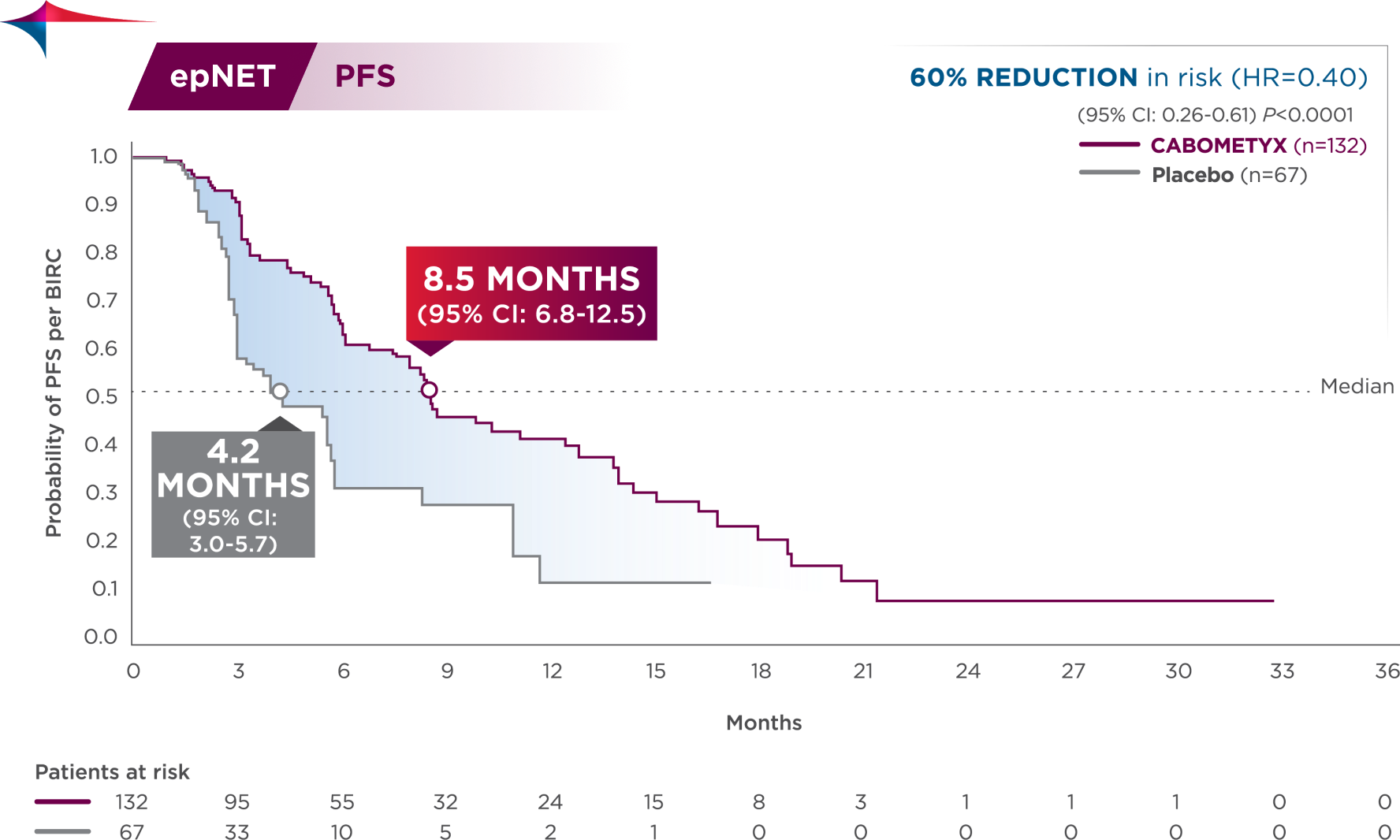
60% reduction in risk of progression or death
PFS results in epNET patients across select disease characteristics2
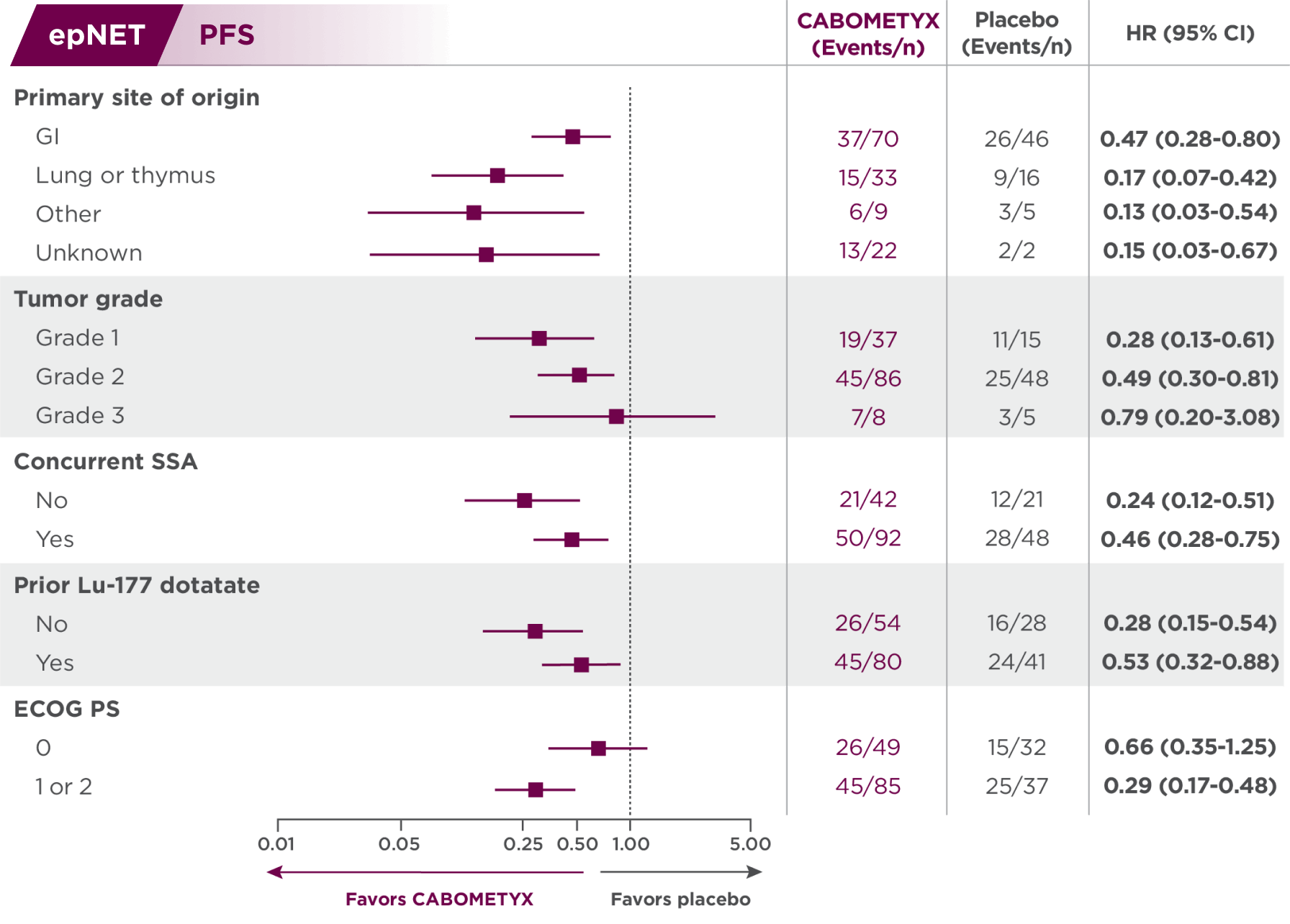
Subgroups were not powered to show differences between treatment arms, and results should be considered hypothesis generating.
Exploratory analysis
PFS results in patients with lung/thymus site of origin3
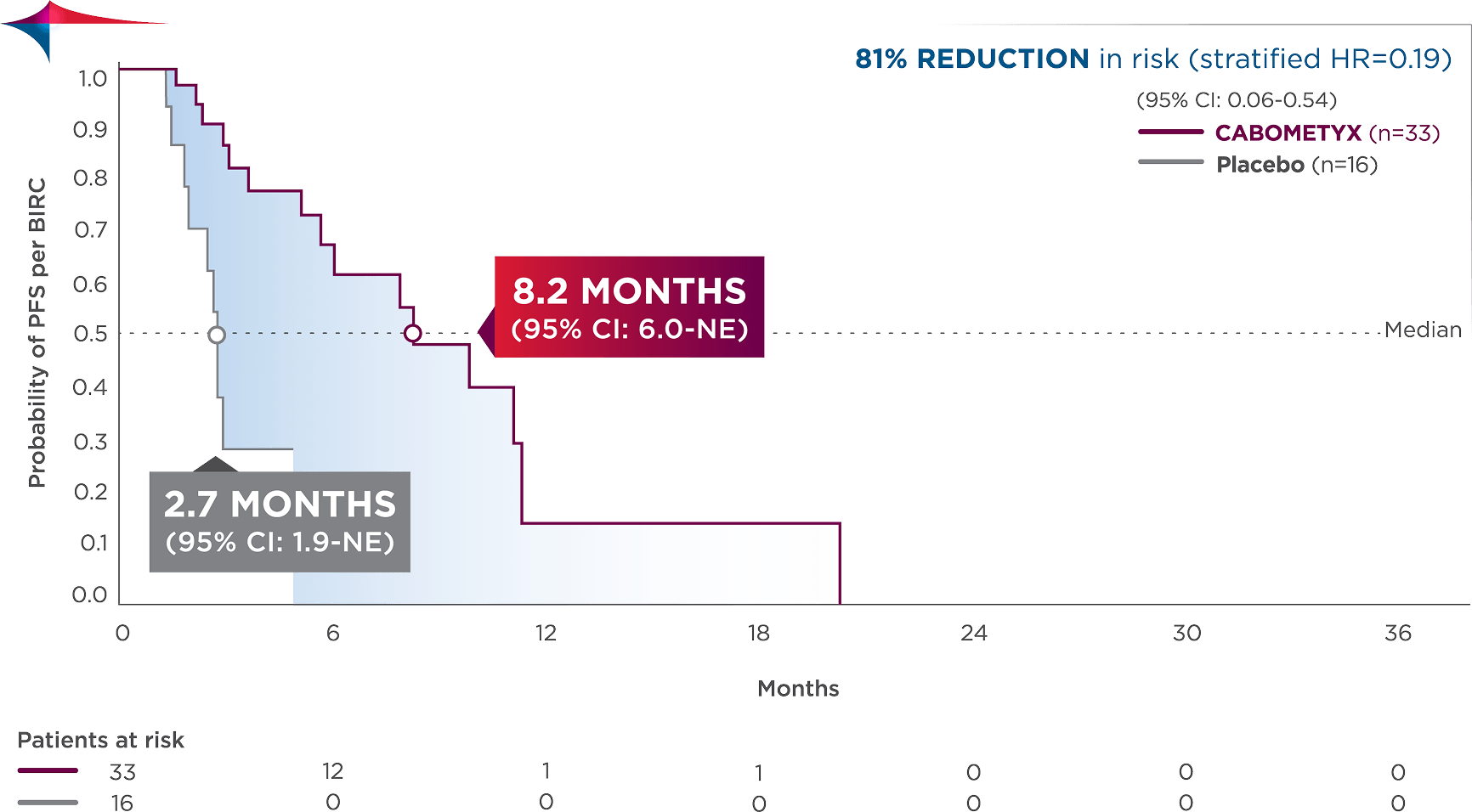
Observed outcomes should be interpreted with caution because of the relatively small subgroup size. Subgroups were not powered to show differences between treatment arms, and results should be considered hypothesis generating.
Exploratory analysis
PFS results in Grade 3 patients across pNET and epNET4
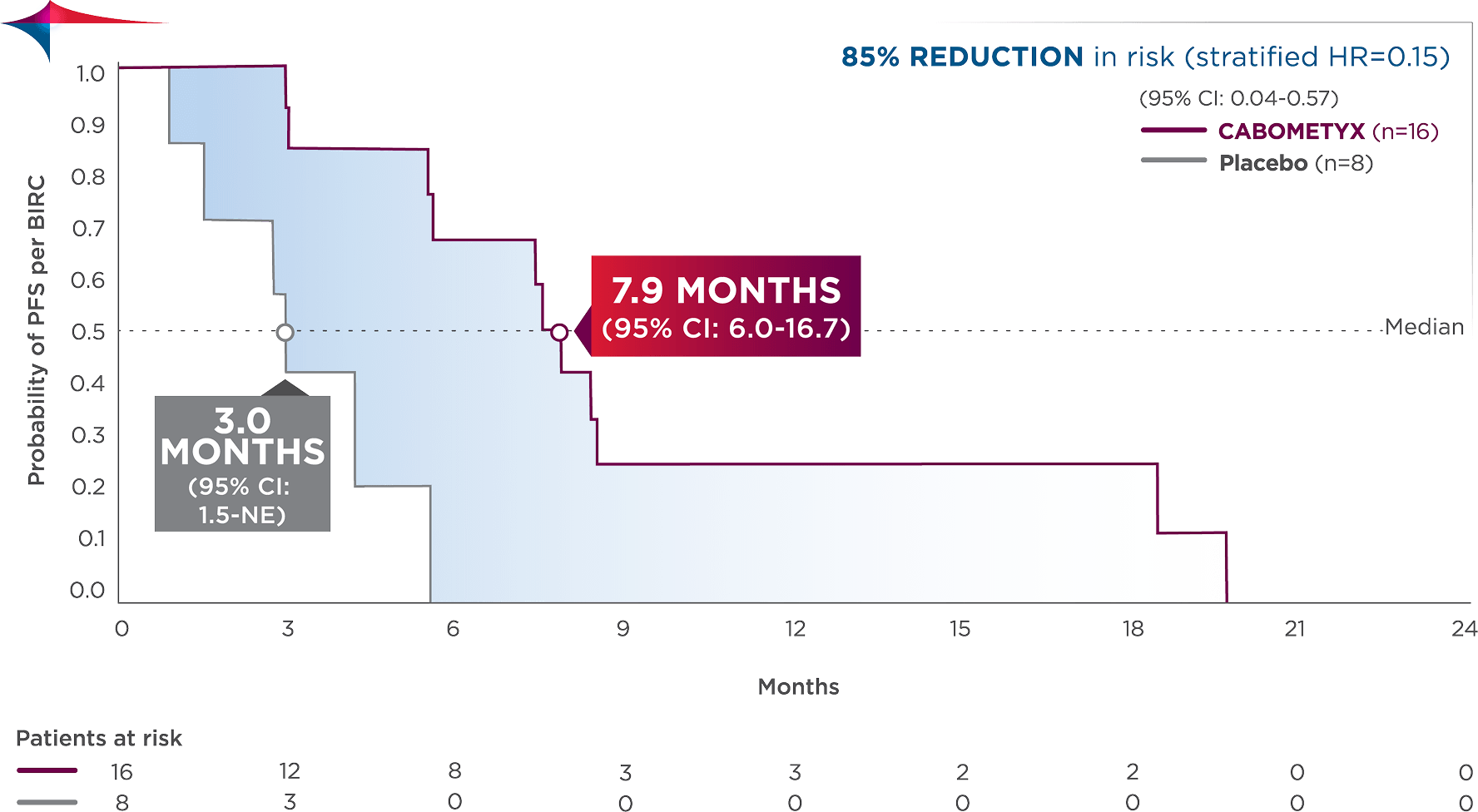
Observed outcomes should be interpreted with caution because of the relatively small subgroup size. Subgroups were not powered to show differences between treatment arms, and results should be considered hypothesis generating.
The Grade 3 analysis pooled 24 patients enrolled in the epNET and pNET cohorts of the CABINET trial.
pNET (n=l2): PFS (median): 13.5 months (95% Cl: 7.6-NE) vs 1.5 months with placebo (95% Cl: 0.9-NE); HR=0.00 (95% Cl: 0.00-NE).
epNET (n=l 2): PFS (median): 6.5 months (95% Cl: 3.0-NE) vs 4.2 (3.0 -NE) with placebo (95% Cl: 0.3-NE); HR=0.45 (95% Cl: 0 .09-2.32).
NE=not estimable.
Descriptive analysis
Tumor response with CABOMETYX in epNET1,5
epNET
CABOMETYX achieved 69% disease control
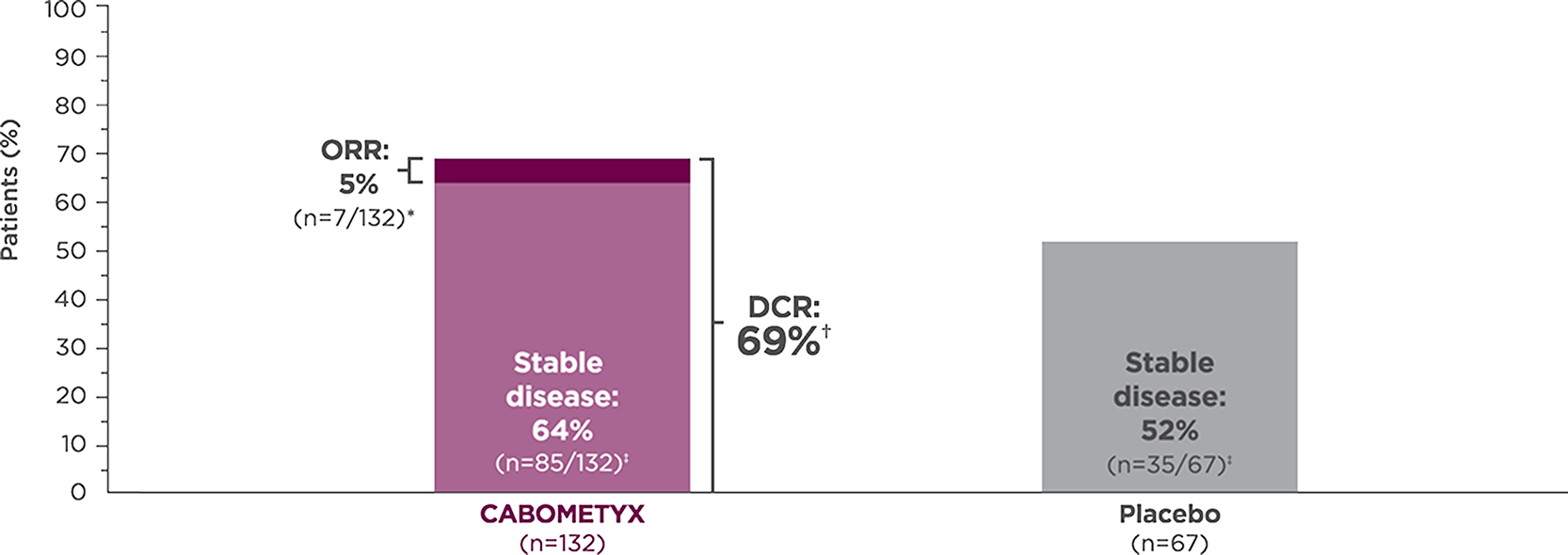
- *
-
All responses confirmed were partial responses.2
- †
-
Disease control rate is defined as the percentage of patients with a complete response, partial response, or stable disease, as measured by RECIST v1.1.5
- ‡
-
Stable disease is defined as neither sufficient shrinkage to qualify as partial response nor sufficient increase to qualify as PD.6 Stable disease may reflect the natural history of disease rather than any effect of the drug.
Descriptive analysis
Reduction in target lesions from baseline2,7
Of the 203 epNET ITT patients, 174 were evaluable (n=111 in the CABOMETYX arm, n=63 in the placebo arm).

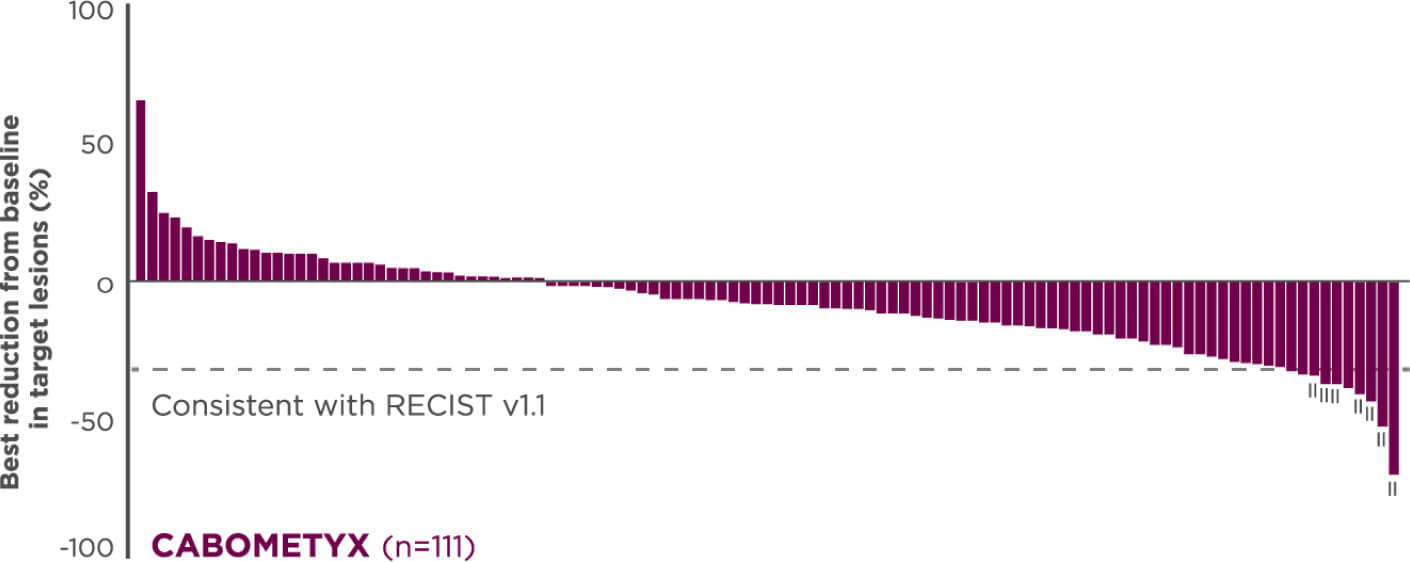
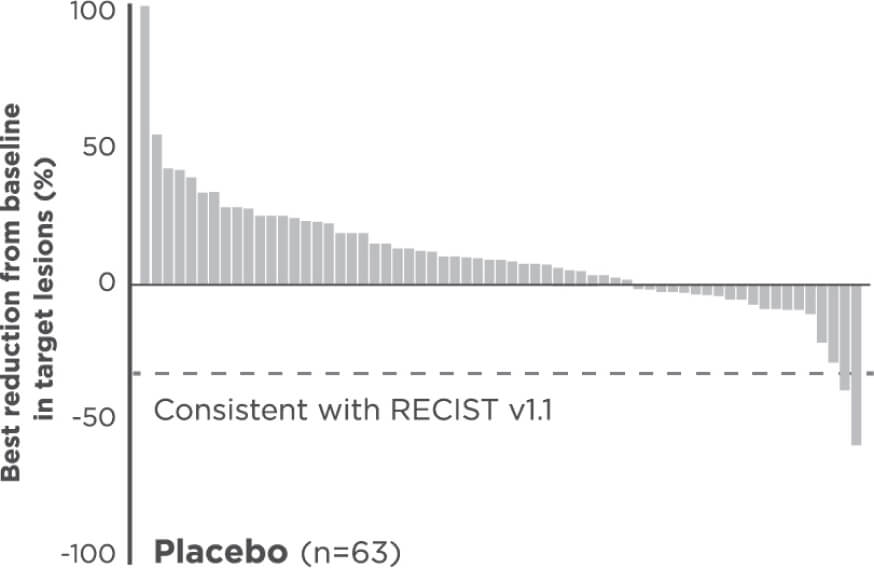
Interpret data with caution.
No formal statistical testing was conducted for the tumor assessment waterfall plots.
Best reduction is maximum reduction in sum of diameters of target lesions. Patients with target lesion at baseline and at least 1 on-treatment tumor assessment are included. Some patients are not represented due to lack of evaluable postbaseline assessment, censoring before first evaluable postbaseline assessment, lack of target lesions, and/or incomplete or inevaluable target-lesion assessment. Data from time points after the first date of any of the censoring events defined for the primary PFS analysis were excluded.5
- §
-
Each vertical line represents 1 patient.7
- II
-
Indicates confirmed partial responses per BIRC.7
Updated analysis
OS results from CABINET1
OS data were not mature at the time of the updated analysis and may be impacted by crossover
epNET
Updated OS
|
63% |
Deaths |
|
60% |
Deaths |
(HR=1.05; 95% CI: 0.71-1.54) |
|
37% of placebo patients crossed over to open-label CABOMETYX |
|||||
|
63% |
Deaths |
|
|
|
|
60% |
Deaths |
|
(HR=1.05; 95% CI: 0.71-1.54) |
|
|
37% of placebo patients crossed over to open-label CABOMETYX |
|
The CABINET trial was unblinded early and patients were allowed to cross over to open-label CABOMETYX regardless of whether they had experienced progression. A later updated OS analysis was conducted when 123 deaths were observed in the epNET cohort.

National Comprehensive Cancer Network® (NCCN®)
Cabozantinib (CABOMETYX) is a recommended systemic anti-tumor therapy for certain patients with:
- Pancreatic NET (Grade 1/2)**
- Gastrointestinal tract NET (Grade 1/2)**
- Lung/thymus NET
- Grade 3 NET**
**Well-differentiated.
NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
2L=second line; BIRC=blinded independent review committee; CI=confidence interval; DCR=disease control rate; ECOG PS=Eastern Cooperative Oncology Group performance status; epNET=extrapancreatic neuroendocrine tumors; FDA=Food and Drug Administration; HR=hazard ratio; ITT=intent to treat; Lu-177=lutetium-177; NE=not estimable; NET=neuroendocrine tumor(s); ORR=overall response rate; OS=overall survival; PD=progressive disease; PFS=progression-free survival; pNET=pancreatic neuroendocrine tumors; RECIST=Response Evaluation Criteria in Solid Tumors; SSA=somatostatin analogue.
References:
- CABOMETYX® (cabozantinib) Prescribing Information. Exelixis, Inc.
- Chan JA, Geyer S, Zemla T, et al. Phase 3 trial of cabozantinib in previously treated advanced neuroendocrine tumors. N Engl J Med. 2024; Published online September 16, 2024. doi:10.1056/NEJMoa2403991.
- Wolin EM, Zemla T, Geyer S, et al. Efficacy and safety of cabozantinib for advanced lung and thymic neuroendocrine tumors after progression on prior therapy: subgroup analysis of Phase 3 CABINET trial (Alliance A021602). Poster presented at: European Society for Medical Oncology Congress: October 17-21, 2025.
- Strosberg JR, Zemla T, Geyer S, et al. Efficacy and safety of cabozantinib for advanced Grade 3 NETs after progression on prior therapy: subgroup analysis of the phase 3 CABINET trial (Alliance A021602). Poster presented at: European Neuroendocrine Tumor Society Conference for the Diagnosis and Treatment of Neuroendocrine Tumour Disease; March 5-7, 2025.
- Data on file. Exelixis, Inc.
- Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228-247.
- Chan JA, Geyer S, Zemla T, et al. Phase 3 trial of cabozantinib in previously treated advanced neuroendocrine tumors [supplementary appendix]. N Engl J Med. 2024; Published online September 16, 2024. doi:10.1056/NEJMoa2403991.
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines·) for Neuroendocrine and Adrenal Tumors V.3.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed October 1, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no representations or warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.


